PURE WATER SUPPLY FOR
PHARMACEUTICAL MANUFACTURERS
This solution brings treatment to water meeting GMP-WHO standard, helps saving energy, ensures stable operation.
In the pharmaceutical industry, clean water is a widely-used raw material in pharmaceutical manufacturing and plays a decisive role for the quality and safety of each product. Therefore, in every pharmaceutical manufacturer, the water treatment system for manufacturing activities must be strictly managed, ensuring simultaneous factors: quality, stable operation.

1. Why do we have to treat water before using it to manufacture pharmaceuticals?
The water used to supply the pharmaceutical industry usually comes from:
- Underground-water
- Water from clean water supply plants in the area
Underground-water sources are often characterized by high amounts of dissolved substances, mainly manganese, calcium, and magnesium, while clean water from water supply plants is not up to the production quality assurance. Therefore, pharmaceutical enterprises are required to treat input water sources to remove impurities or microorganisms that affect the quality of pharmaceuticals.
2. Standard of water supplied for pharmaceutical production activities
Currently, water supplied for pharmaceutical activities in Vietnam is according to GMP standards. Specifically:
| NO | PERAMETER | CONCENTRATION LIMITS | ACCORDING GPM STANDARD | UNIT |
| 01 | PH(25°C) | 5.2-7.0 | WHO | U.S. |
| 02 | TASTE | – | Pharmacopoeia Part III-VN | – |
| 03 | Calcium and
Magnesium |
0 | 0 | PPM |
| 04 | Color
temperature |
0 | Pharmacopoeia Part
III-VN |
Pt – Co |
| 05 | ClorI | 0.5 | 0.5 | WHO |
| 06 | Sulphate | No change in color after
1 hour |
Determined according to Ph standard Eur (Vo. II) | PPM |
| 07 | Oxidants | No change in color after 5
minutes |
Ph.Eur | No change in color after 5 minutes |
| 08 | Ammonium | 0.2 | Ph.Eur | PPM |
| 09 | Nitrate | 0.2 | WHO | PPM |
| 10 | Aluminum | 0.01 | Ph.Eur | PPM |
| 11 | Heavy metal | 1 | Ph.Eur | PPM |
| 12 | Evaporation
sludge |
0.001 | Ph.Eur | % |
| 13 | Conductivity | 1.3 (Water for injection) | WHO | mS/cm
(25oC) |
3. Classification of water used in the pharmaceutical industry:
Normally, the water used in pharmaceutical industry is classified into 04 Categories for different uses:
Domestic water that is municipal tap water, used for dining area, toilets,
watering plants outside the production area Demineralized water that has undergone soft water treatment, removed some impurities ions in water and some microorganisms, mainly used for boiler water, autoclaves for injections – eye drops, rinsing bottles for eye drops, syringes, machines …
Pure water that is used to prepare products that do not require sterility and are free of fevercausing substances Distilled water
4. What factors should a pharmaceutical manufacturer consider when investing in a water treatment system?
Currently, the technological process of water treatment for pharmaceutical manufacturing needs to ensure (03) the following factors:
- Water quality
- Easy to operate Saving operating costs
In addition, businesses also need to consider a number of other factors:
- Requirements for machinery and equipment assurance, with clear
- Analysis, survey of input water before treatment for production
- Careful analysis of the use needs for each stage of pharmaceutical manufacturing, to make appropriate treatment plans
- Consider long-term economic performance, including operation, maintenance, and labour
- Supply water treatment solution provided by Toptech Enterprse:
Technological advantages:
- Concentrated water from the filtration system can be reused, without oxidation risk for the RO filtration stage from oxidizing
- Low- Power
- Water meets GPM-WHO standard
6. Example of water treatment process meeting GMP standard:
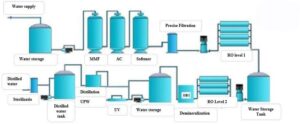
Water treatment process meets GMP-WHO standard for reference The treatment process consists of the following main components:
- Multi-material pretreatment system: removes sediment and suspended particles in water, and absorbs free chlorine, to prevent oxidation of free chlorine while
avoiding damage to RO membrane at Post-processing step. The material can also adsorb heavy metals, small organic molecules. After this step, the turbidity in the water is controlled at SDI ≤ 5.
- 2-stage RO filter system: The reverse osmosis device has the advantages of compact structure, small area, high production volume and low energy Reverse osmosis equipment is the most important desalination equipment in this process, including safety filter, phase I pump, phase I RO system, phase II pump, phase II RO system. It also help eliminate bacteria, virus and heat source.
- Demineralization: Urban water contains sodium, calcium, magnesium, chloride, nitrate, bicarbonate, silica and other soluble These salts include anions and cations. More than 99% of ions can be removed by appropriate reverse osmosis (RO). Municipal water also contains trace metals, dissolved gases (such as CO2) and other weak ionic compounds that must be removed in industrial applications (such as boron and silica).
- The conductivity of water after the RO filtration step (the input water of the EDI demineraliser) is usually between 4 and 20 us / cm, ie the reaction rate is 50-250 kΩ.cm; and according to different application fields, the resistance of pure or deionized water is usually from 2-18.2 MΩ. Demineralization process is to remove unwanted ions through hydroxyl ion exchange or hydroxide
- Cleaning chemical system: including chemical containers, dosage adjustment equipment, agitator for routine RO
- Automatic control system
If your business is interested in solution to inlet water treatment, or has a demand for ion exchange materials, please contact Toptech Enterprise via phone number +8801733337770 or email:- toptechbd@yahoo.com for direct consultation.
PDF :LINK: PURE WATER SUPPLY FOR PHARMACEUTICAL MANUFACTURERS